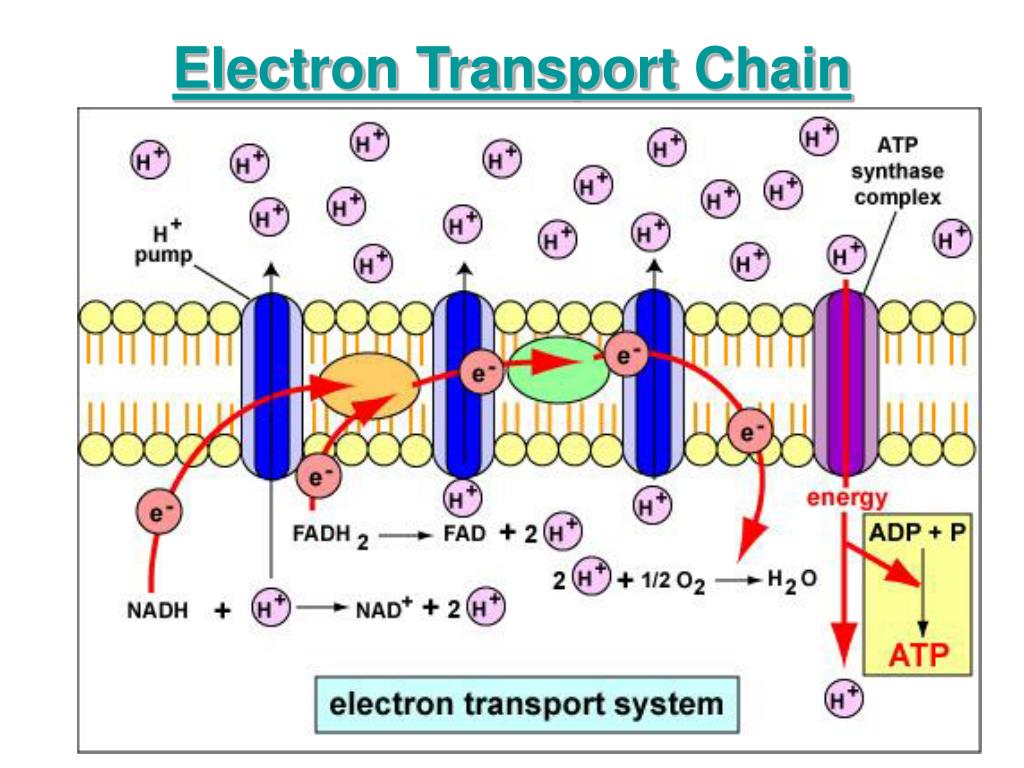
The electron transport chain creates a proton gradient. This energy is used to pump protons across the mitochondrial membrane. Read also: 18 Captivating Facts About Neurotransmitter Electrons move through the protein complexes in a series of redox reactions.Īs electrons pass through the protein complexes, they are transferred from one molecule to another, resulting in a release of energy. 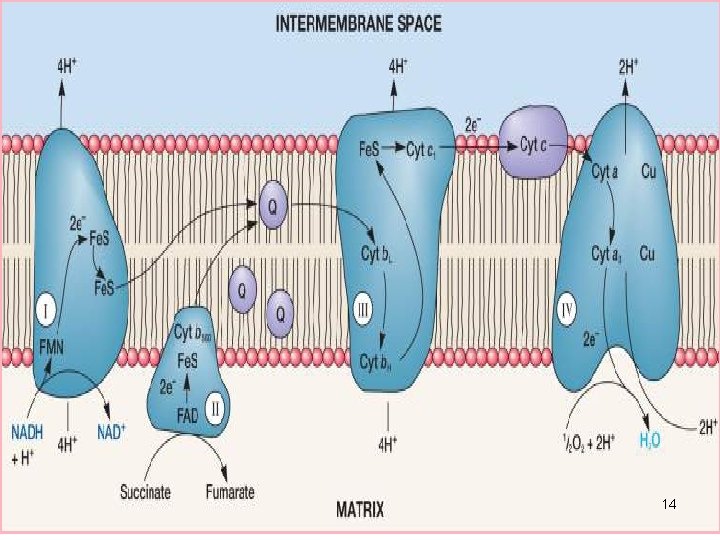
NADH and FADH2 are high-energy molecules that donate electrons to the electron transport chain, initiating the flow of electrons through the complexes. It relies on electron carriers such as NADH and FADH2. The four protein complexes involved in the electron transport chain are complex I (NADH dehydrogenase), complex II (succinate dehydrogenase), complex III (cytochrome bc1 complex), and complex IV (cytochrome c oxidase). The electron transport chain is composed of four protein complexes. It plays a crucial role in producing adenosine triphosphate (ATP), the energy currency of cells. The electron transport chain is a series of protein complexes located in the inner mitochondrial membrane. CC BY 4.0.The electron transport chain is an essential process in cellular respiration. Grey, Kindred, Figure 4.14 Overview of the electron transport chain (ETC). Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins, 2018, Chapter 2: The Fed or Absorptive State, Chapter 19: Basic Concepts of Regulation: Section IV.A.1.2, Chapter 20: Cellular Bioenergetics, Chapter 22: Generation of ATP from Glucose: Section I.A.B.C, III, Chapter 24: Oxidative Phosphorylation and the ETC: Section I.E, II, III, Chapter 31: Synthesis of Fatty Acids: Section I.A.B, IV, V. Marks' Basic Medical Biochemistry: A Clinical Approach, 5th ed. Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins, 2017, Chapter 6: Bioenergetics and Oxidative Phosphorylation: Section V, VI, Chapter 8: Introduction to Metabolism and Glycolysis, Chapter 9: TCA Cycle and Pyruvate Dehydrogenase Complex: Section IIA, IIB, Chapter 11: Glycogen Metabolism: Section V, VI, Chapter 16: Fatty Acid Ketone Body and TAG Metabolism: Section II, IV, V, Chapter 23: Metabolic Effect of Insulin and Glucagon, Chapter 25: Diabetes Mellitus. Lippincott Illustrated Reviews: Biochemistry, 7th ed. There are site specific inhibitors of the ETC to be aware of, and these will disrupt electron flow reducing overall ATP production.įigure 4.14: Overview of the electron transport chain (ETC).įerrier, D. Electrons are passed down an electrochemical gradient, and molecular oxygen is the final electron acceptor (molecular oxygen). Complex II (succinate dehydrogenase) is not required for oxidative phosphorylation because it does not span the mitochondrial membrane (figure 4.14). Complex I and Complex II function in parallel (rather than series) with each other having preference for NADH or FADH\(_2\), respectively. With the exception of CoQ all proteins are bound to the mitochondria membrane, and electrons are passed between metal containing cytochromes. The process of oxidative phosphorylation (figure 4.14) involves the coupling of electron transfer with the pumping of protons to generate an electrochemical gradient across the mitochondrial membrane. As the electrons are passed down their oxidation gradient, some of the energy is lost, but much of this energy is used to pump protons into the intermembrane space of the mitochondria. In the production of NADH and FADH\(_2\) by the TCA cycle, \(\beta\)-oxidation or glycolysis is funneled directly into the electron transport chain (ETC) where each of these reduced coenzymes will donate two electrons to electron carriers.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
